WP2 - Identification and validation of new autoantibodies as biomarkers
Leader : L. Komorowski (PhD, Euroimmun, Lübeck-Germany)
EUROIMMUN and J.Honnorat's lab have already identified new autoantibodies associated with AE or PNS. The diagnostic value of these newly identified autoantibodies as well as of already known autoantibodies will be evaluated by EUROIMMUN and corresponding in-vitro diagnostic tests will be developed in order to be commercialized.
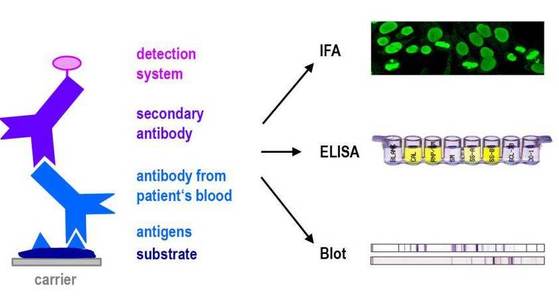
The aim of this WP is to use the biological samples collected in WP1 to identify new autoantibodies and to validate their clinical values and to use the data of WP3, WP4 and WP5 to test the presence of autoantibodies against suspected targets.
Task 2.1: Validation of EUROIMMUN new autoantibodies
Objective: Using the biological samples collected in task 1.1 to 1.3, the diagnostic value of these newly identified autoantibodies as well as of already known autoantibodies will be evaluated by EUROIMMUN and corresponding in-vitro diagnostic tests will be developed. The diagnostic performance of these prototypic tests will be determined in order to commercialize them.
Task 2.2: Characterization of antigens targeted by new autoantibodies
Objective: Characterize the antigens recognized by these autoantibodies.
Task 2.3: Improvement of existing tests
Objective: Improve diagnostic sensitivity and specificity of tests already in the market for the detection of autoantibodies associated to PNS and AE.
Task 2.4: Re-assessment of diagnostic strategies
Objective: Improve the current accepted tests strategy in order to minimise the lead to a false negative serological result that may overlook PNS patients.
Task 2.5: Screening of potential new targets to identify new potential autoantibodies as biomarkers
Objective: Use up to 50 candidates identified in WP3, 4 or 5 to be produced as recombinant proteins for screening blood samples of patients with suspected or proven PNS or AE.
Task 2.6: Development of diagnostic kits
Objective: Develop diagnostic kits for the detection of the new biomarkers identified in task 2.1 to 2.3 for commercialisation.


